Kinetic energy is a fundamental piece in the concepts of physics. Kinetic energy indicates the energy an object has because of its motion. This form of energy gets found in everything from nature to artificial circumstances. First, however, let us look at the mathematic components and discover whether kinetic energy can become negative.
Beautifully put by a review done by the Khan Academy, kinetic energy cannot be negative, but the fluctuations in direction can be negative. This idea means the energy itself can never be a negative value, but where the force is traveling can depict its negative effect.
Because this ideology is so important in modern development, kinetic energy gets taught at a young age. Below are certain topics that one should consider when studying kinetic energy:
- The functioning of kinetic energy
- Formulas used to calculate kinetic energy
- Interesting concepts about kinetic energy
- Certain mistakes and false news around kinetic energy
- The special connection between kinetic energy and potential energy
Now, let us look at the points listed above individually and learn a more in-depth knowledge on these various topics to help better understand the kinetic energy ideology.
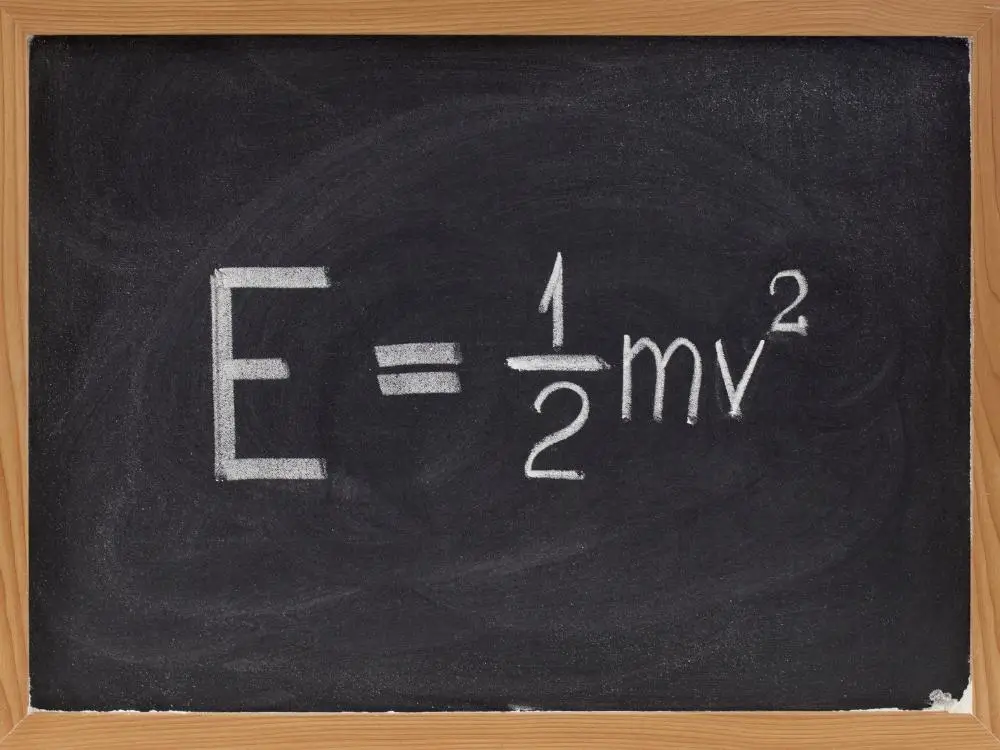
How Does Kinetic Energy Work?
If we wish to hasten an object, we must first initiate a force. Applying force entails us doing some extra work. After the additional work gets done, energy has successfully been transferred to the object, hence concluding to the thing moving with a newly found constant speed.
This transferred energy is known as kinetic energy, and its value depends on the mass and speed achieved. In addition, kinetic energy can transfer between separate objects and become transformed into other types of energy.
In other words, it is energy from translational or rotational motion – SI units of joules (J). Translational motion is a linear motion of a rigid body – SI units of ms.
In addition, translational kinetic energy (K) is energy due to the linear motion of a rigid body – SI units of joules (J).
How Is Kinetic Energy Calculated?
To calculate kinetic energy, one must follow the structured formulas and begin by finding variables such as work done, indicated as W, and force, shown as F.
While collecting all the information you require, think of a box of mass, the symbol being m, moved at a specific distance, the sign being d, along a surface by a force parallel to that particular surface.
Here is how the variable W gets calculated.
W = F ⋅ d
= m ⋅ a ⋅ d
If we look back to the kinetic equations of motion, we know that we can input the acceleration only if we know the initial and final velocity, vi and vf, and the distance. Below is the substitute equation for calculating the work done, W.
W = m ⋅ d ⋅
= m⋅
= ⋅ m ⋅ vf2 − ⋅ m ⋅ vi2
Therefore, when a certain amount of work gets done, the value is . m. v2, which we call kinetic energy and indicated by K, changes. Alternatively, the outcome could indicate that the change in kinetic energy equals the amount of work completed on an object or system.
Wnet = ΔK
These outcome results are known as the work-energy theorem and apply to many different things, even with forces that change in direction and magnitude. Understanding this idea is vital in studying energy conservation and conservative forces.
Additional Equations Related To Kinetic Energy
Translational kinetic energy is instantly proportional to mass and the square of the magnitude of the velocity (speed). Therefore, the symbol K is translational kinetic energy, m is mass, and v is the magnitude of the momentum.
K = mv2
Changes within kinetic energy are the difference between final and initial kinetic energy (velocity). ΔK symbolizes the change in kinetic energy, and v and v0 are the magnitudes of the last and initial rate.
ΔK = m(v2 – v20)
Some Interesting Facts About Kinetic Energy
There are a few exciting things about kinetic energy that we can discover from the equations shown above. Firstly, kinetic energy depends on the velocity of the objects squared. This concept indicates that kinetic energy will quadruple when a specific object’s momentum doubles.
For example, a car moving at 60 mph has four times the kinetic energy of an identical vehicle traveling at 30 mph, resulting in a higher probability of death and destruction in the event of a crash.
Kinetic energy must be either zero or a positive value. So, for example, while the velocity of an object can have a positive or negative value, speed squared will always result in a positive figure.
In addition, kinetic energy is not a vector, and to better understand this meaning, we’ll give an example. A tennis ball thrown to the right with a velocity of 5 m/s has the precise same kinetic energy as a tennis ball thrown down at a speed of 5 m/s.
Common Misconceptions And Mistakes About Kinetic Energy
Firstly, kinetic cannot and will never be a negative value, although the change in kinetic energy ΔK can be negative.
This conclusion comes about because mass cannot be a negative figure, and the square of speed will always leave you with a positive number, further proving that kinetic energy can’t be negative.
Either an object is moving and has positive kinetic energy, or the thing is not moving at all, leaving it will produce kinetic energy with the value 0.
If the final speed is less than the initial speed, then the last kinetic will be less than the initial kinetic energy, and ΔK will become a negative value.
Secondly, some individuals believe that doubling speed means double the energy output. The kinetic energy corresponds to the square of the speed, so double the rate increases the kinetic energy force by a factor of 4. However, doubling the speed does, in fact, double the momentum.
The Connection Between Kinetic And Potential Energy
One deals with potential or kinetic energy whenever one uses or stores energy. Therefore, let us explore the relationship between these two forms of energy.
As stated previously, energy is an intriguing concept. It is an element that can neither be created nor destroyed; however, one can alter it.
Professionals know that potential energy is position relative and kinetic energy is motion relative. Therefore, the primary relationship between the two energies is their ability to convert into each other.
In other words, they can constantly transform into each other back and forth, resulting in a repetitive cycle.
What Is The Difference Between Potential And Kinetic Energy?
Your ability to do any work is energy because you need the energy to do any work. This saying means that potential and kinetic energy are two forms of energy that can convert into each other.
Kinetic energy can convert to potential energy and vice versa. Because we have already addressed the definition of kinetic energy, let us look at the meaning of potential energy.
Potential energy is the stored energy within any object or system by principles of its position or arrangement of parts.
In addition, the environment outside cannot influence the object or system, such as height or air.
Stationary Vs Moving Objects
Different from potential energy, the kinetic energy of a specific item is relative to other stationery or moving objects present in that environment. For example, the kinetic energy of an object will be higher if the object gets situated at a higher height.
More simply put, potential energy is not transferrable, and it purely depends on the height or distance and mass of the item in question.
On the other hand, kinetic energy can be transferred from one object to another via vibration and rotation and is wholly dependent on an object’s speed, velocity, and mass.
Is A Battery Kinetic or Potential Energy?
A battery is an element of potential energy.
We will have to get a little technical to understand this saying further. The battery contains potential electrical power when the electrons move from the cathode to the anode – when a battery is charging.
When the electrons adjust to move in a different direction, the battery converts this chemical potential energy to electricity in the circuit, hence discharging the battery.
So, in conclusion, a battery consists entirely of potential energy.
Is Electrical Energy Potential Or Kinetic Energy?
Electrical can either be kinetic or potential energy because it gets created from an electric charge flow.
So again, referring back to the example of a battery, we have discovered that it has electrical potential energy while charging. But the minute you apply force to the battery, the charged particles start to work, converting the potential energy to kinetic energy.
A process similar to this happens when switching on a light causing potential energy to travel down wiring and continue to convert into light and heat that consists of both forms of kinetic energy.
Is Sound Energy Kinetic Or Potential Energy?
Like electrical energy, sound energy can be a form of potential energy and kinetic energy. However, before we get into any specifics, let us understand the definition of sound energy.
Sound energy refers to the energy that emits from vibrating objects. The sound often gets pictured as a wave that travels through a medium, for example, air, which allows it to contain potential energy and kinetic energy.
For instance, when one plays an instrument such as the drums, they vibrate due to sound waves emitting from the striking of the drums. These sound waves travel and oscillate, continuing to create kinetic energy.
However, when the drums remain left untouched, they will primarily consist of potential energy since the instrument is not in motion and gets left with the potential to make a sound.
Is Thermal Energy Kinetic or Potential?
Thermal energy is the more formal name to describe heat energy. One can find both potential and kinetic energy within thermal energy.
We can look at the example of boiling water to understand this concept. Water, when placed on the stove, has potential energy. However, when it starts to heat up, the water molecules rapidly move, creating kinetic energy.
Remembering back to science lessons in school, the electrons of an atom constantly have potential energy. When you place pressure on the electrons, they will begin to move faster, colliding with each other and emitting thermal energy as heat.
In other words, thermal potential energy is potential energy at the atomic and molecular levels when particles showcase the potential of transforming into kinetic energy.
On the other hand, thermal kinetic energy occurs when the atoms and molecules shift due to heat and temperature.
Is Radiant Energy Kinetic Or Potential?
Scientists state that radiant energy is a form of kinetic energy created when electromagnetic waves move through space.
A new fact that you might learn right now is that the sun is one of the biggest radiant energy sources on our entire planet. Think back and remember when you got that warm feeling when you stepped out of the sun. That is the sun’s radiant energy touching your exposed skin.
More precisely, the electromagnetic waves cause our skin’s molecules to move faster, leading to the creation of kinetic energy.
Reasons Why Kinetic And Potential Energy Are Indispensable
One cannot disagree that potential and kinetic energy are an indispensable part of our lives—all of the simple little tasks that we do involve both forms of energy.
Of course, as you have learned, there are many energies, such as thermal, sound, and electrical energy. However, these energies have something in common: either potential or kinetic energy.
Potential and kinetic energy also play a vital role in making our earth greener as they assist in creating renewable sources of energy, such as using energy from wind or water.
In other words, we will always deal with these energy forms because energy is a constant element that will never be lost. It does not disappear; however, it does change.
Resources
- https://www.khanacademy.org/science/in-in-class11th-physics/in-in-class11th-physics-work-energy-and-power/in-in-class11-kinetic-energy-ap/a/kinetic-energy-ap1
- https://justenergy.com/blog/potential-and-kinetic-energy-explained/#:~:text=You%20now%20know%20that%20potential,energy%2C%20and%20then%20back%20again
- https://justenergy.com/blog/potential-and-kinetic-energy-explained/



