Heat pumps make use of the natural property of the refrigerant medium to absorb heat when it boils or evaporates. Heat exchange is a natural phenomenon for all liquids when evaporating into a gas. When water absorbs enough heat, it will start to boil as the liquid water escapes the surface tension of the liquid to become a gas or water vapor.
Liquid to gas phase changes that naturally occur below room temperature is ideal for heat pumps. Little or no energy is needed to let the refrigerant change from liquid to gas. The refrigerant absorbs the heat and can thus cool the surrounding. It pumps the heat out of the room.
The principle of a heat pump is used in air conditioning systems, refrigerators, and water heaters to transfer heat away from or towards an environment. We will look at some everyday applications and an example of how nature works.
- How a heat pump can be used to heat during winter and cool during summer
- How domestic refrigerators work
- How is the efficiency of a heat pump determined
- How the heat pump principle works in nature
Heat pumps are systems that absorb kinetic energy from one medium and transfer it to another medium due to the natural properties of the refrigerant medium. Let’s investigate the heat pump mechanism more closely.

The Physics Of Temperature And Energy
The definition of temperature is that it is a measure of the amount of Kinetic energy in the surrounding medium. The mercury in a thermometer absorbs the heat from the air or liquid into which the thermometer is placed.
- The Kinetic energy of the molecules moving about is transferred from the air molecules to the mercury atoms.
- The increase in the vibrational energy (kinetic energy) of the mercury atoms in the thermometer bulb and tube will expand the volume of the mercury.
- The expanding mercury will push up the thermometer tube and reflect the heat of the surrounding medium.
Calibration of the thermometer requires that the bulb of the thermometer is held in ice water to establish the zero Celsius point and in boiling water to establish where a hundred degrees Celsius is.
The transfer of kinetic energy from one medium to another is used in heat pump mechanics.
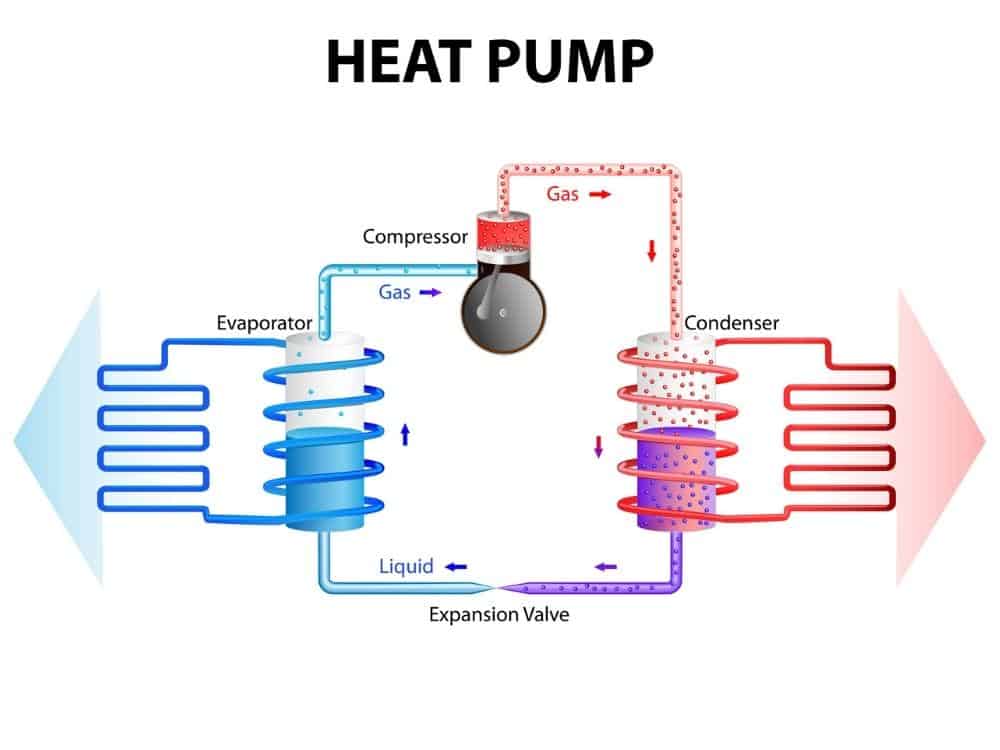
The Components Of A Heat Pump And How Does It Work?
A commercial heat pump system, as is found in modern refrigerators, ice makers, water heaters, and air conditioners, all have the following components:
- A refrigerant agent such as Carbon Dioxide or Freon
- A compressor and electric motor
- An electric fan blower
- A condenser coil
- A radiator coil
- Non-return valves
- Expansion valves
Compressor Pump
The compressor pump receives cool, low-pressure refrigerant gas in the summer and pumps it to a high-pressure routing valve into the high-pressure condenser coil.
An electric fan draws air over the condenser coil drawing heat from the metal surface of the condense coil.

Pressurized Gas
The pressurized gas is thus cooled and, under pressure, becomes a cold liquid. The cold pressurized liquid passes through the first expansion valve (unidirectional check valve). The cold liquid is still under pressure as it flows through a second expansion valve.
Liquid
As the liquid passes through the second expansion valve, it goes from the high-pressure loop into the low-pressure loop.
The drop in pressure causes the liquid to boil and become a gas as it moves through the evaporator coil inside the house. The heat absorbed from the air inside the house cools the interior air in the house down.
Absorbtion
The gas absorbs the heat as it flows back to the compressor to start the entire cycle over again. The system thus absorbs heat from the inside of the house and pumps it to the outside of the house by using the refrigerant medium to “pump” the heat out of the house.
The refrigerant is always at a constant volume in the sealed system of pipes. Unless a leak develops, causing the gas to escape from the system, the system can operate indefinitely.

Leaks
Unfortunately, the seals in the compressor pump and the pipe joints of the system are not perfect and do develop leaks over time. The constant cycling of high and low temperatures can also cause mental fatigue in the pipes, which will result in leaks over time.
Winter Mode
In winter mode, the heat pump system will reverse direction and, using the same principles as explained before, will absorb heat from the outside ambient conditions and pump the heat to the inside of the house.
Instead of cooling the house interior, the same system, when run in reverse, will heat the inside of the house.
In the winter cycle, the air outside may be cold enough to freeze water, but it is still high enough to make the refrigerant boil and turn from a liquid to a gas. The problem is that the moisture in the air freezes up on the outside evaporator coil. The ice on the coils acts like an insulator, preventing the gas from absorbing heat.
This problem is easily overcome by programming the electronic control system to run the system in summer mode for short periods. The defrost cycle allows the heat from inside the house to melt the frost and ice from the external evaporator coil.
Once the evaporator coil is free of ice, the heating cycle is automatically started again, and the house is warmed on the inside.
If the defrost cycle does not run for long enough, the frost and ice build-up will become excessive and require an adjustment to lengthen the defrost cycle.

How Does A Refrigerator Heat Pump Work?
The modern domestic refrigerator is a very efficient heat pump built onto a well-insulated box, fitted with a door to insert or remove items from the box. The most efficient refrigerators are upright, with a lid opening upwards. As cold air is heavier than warm air, cold air will always fall to the bottom of the fridge.
In an upright domestic fridge, you will feel the cold on your feet when you open the fridge’s door as the cold air falls out. Opening the fridge’s door too much makes it lose cold air and requires the compressor and cooling fan to run more often.

Copper Tube
In the insulated box of the fridge, a copper evaporator tube is routed along the top of the back wall of the box. The refrigerant gas flowing through this tube will absorb heat from the air inside the fridge and transfer it via the condenser coil on the outside of the fridge to the outside air.
Suppose you place your refrigerator too close to the wall and insufficient space around the condenser coil to dissipate the heat being pumped out of the fridge. In that case, the system will have to work harder and use more power to generate sufficient cooling.
If the fridge’s interior is not getting cold enough, the first issue to check on is whether the condenser coil is clean of dust and far enough from the nearest wall to allow sufficient airflow.
Condenser Coil
The warmed gas from the fridge’s interior is circulated via the external condenser coil to cool down before the compressor pumps it into the high-pressure loop of the fridge, where it is liquefied under pressure once again. The liquefied refrigerant will flow back into the fridge via an expansion valve.
Once the liquid moves past the expansion valve into the low-pressure loop of the evaporator, the liquid boils and becomes a gas.
This cold gas will absorb heat from inside the fridge and move the heat to the outside of the fridge again to escape via the condenser coil.

How Energy Efficient Is A Heat Pump?
The Coefficient of Performance (COP) of a heat pump gives you a measure of the efficiency with which the system will extract kinetic energy from the outside ambient air to a refrigerant and then transfer that energy to the air inside the house.
To compress the refrigerant, energy must be supplied to a compressor pump forcing the refrigerant gas to condense.
The Coefficient of Performance measures how much heating power is achieved for the amount of power needed by the compressor unit to generate that heat.
- A COP = 2 means that 2 kW of heating power was generated while the compressor needed 1 kW of power to condense the refrigerant gas during the cycle.
- A COP of 1 or less means that the system is not efficient. A COP above 3 is considered very efficient, and a COP of 4.5 is considered the best possible COP achievable in practical applications.
Modern heat pumps use a chemical compound called a refrigerant that functions well as it only requires a small amount of energy to change from a liquid to a gas and back again.
This compound is called Freon, and the most common blend used in heat pumps today is designated as R410A.

Natural Heat Pumps In Nature
The best example of a natural heat pump is the ocean’s effect on the temperature of the coastal areas. In nature, the refrigerant medium is the air and moisture in the troposphere. The sun warms the ocean’s surface and heats the molecules in the upper layers.
The water evaporates from the liquid ocean causing the water to cool and the hot vapor to rise into higher elevations above the ocean surface. As the warm moist air rises, it will condense and form clouds. The clouds will gown in density as the warm moist air accumulates.
In the upper layers of the atmosphere, the colder layer of cold air will cool the water vapor down and condense into droplets of water. When a sufficient amount of water droplets have formed in the clouds, a saturation point is reached, and the droplets will start to fall to the surface of the earth again.
The Water Cycle
This natural cycle is referred to as the water cycle, and without it, life on earth would not be possible. The water cycle helps regulate the planet’s temperature and ensures that moisture is carried to the arid inland areas where it will cause rain or snow at higher altitudes.
The water vapor in the atmosphere also created a natural hothouse effect around the planet, making the ambient conditions on earth about thirty degrees Celsius higher than what is theoretically possible just due to solar heating.
People prefer living in Florida as opposed to Arizona because of the oceanic heat pump that makes the climate in Florida humid but cool as opposed to the arid dry heat of Arizona.
The other natural heat pumps around the planet are the tropical forests. In the Amazon jungle, the trees produce large amounts of oxygen and moisture through photosynthesis.
These lush green forests produce a huge amount of water vapor that rises and transfers heat away from the equatorial regions to the temperate zones.
Tropical Rain Forests
The destruction of tropical rain forests is one of the biggest reasons for destabilizing the water cycle and the amount of Carbon Monoxide in the atmosphere. Carbon Monoxide and other gasses like Methane absorb the heat from the sun far better than oxygen and nitrogen.
The net effect is that as the levels of carbon monoxide, carbon dioxide, and Methane increase due to human activity on earth, the earth’s average temperature has increased. The heat is pumped from the excessive solar energy absorption to the oceans and the polar ice caps.
This effect is called global warming, and the result of an increase of two to five degrees Celsius throughout the globe will make life on earth very difficult.
Severe weather events have become more evident in the past fifty years and will intensify and cause more destruction of natural systems on earth.
References:
- https://youtu.be/14MmsNPtn6U – How Does A Heat Pump Work
- https://c03.apogee.net/mvc/home/hes/land/el?utilityname=peco2&layout=IFrame&spc=hel&id=1804
- https://purethermal.co.uk/natural-heat-pumps/
- https://climate.nasa.gov/resources/global-warming-vs-climate-change/#:~:text=Global%20warming%20is%20the%20long,gas%20levels%20in%20Earth’s%20atmosphere.




